GFP-Trap Magnetic Particles M-270 Affinity Reagents
For Super-Sized GFP-Fusion Proteins and Large Binding Complexes
Analyze the GFP fusion proteins – in a tube
Fluorescent fusion proteins are popular tools to study protein localization and cellular dynamics. These constructs usually fuse the entirety–or at least a functional domain–of a target protein to one of the many types of reporter fluorescent proteins. Those originally derived from the jellyfish A. victoria are referred to as green fluorescent protein, or GFP. It’s easy to image cells using a GFP-fusion protein construct, though to get the full picture such imaging data is often combined with additional biochemical information for the protein (or protein domain) of interest. For such biochemical experiments, typically a second fusion protein is designed using a different “tag” domain that allows for purification. These additional in vitro analyses can be used to confirm functionality of the “tagged” fusion construct, as well as to pull out multi-protein complexes that may form in the cellular milieu. The lack of specific, reliable, and efficient reagents has limited the use of GFP, and related fluorescent fusion proteins, for use in both cell biology studies and direct biochemical analysis.
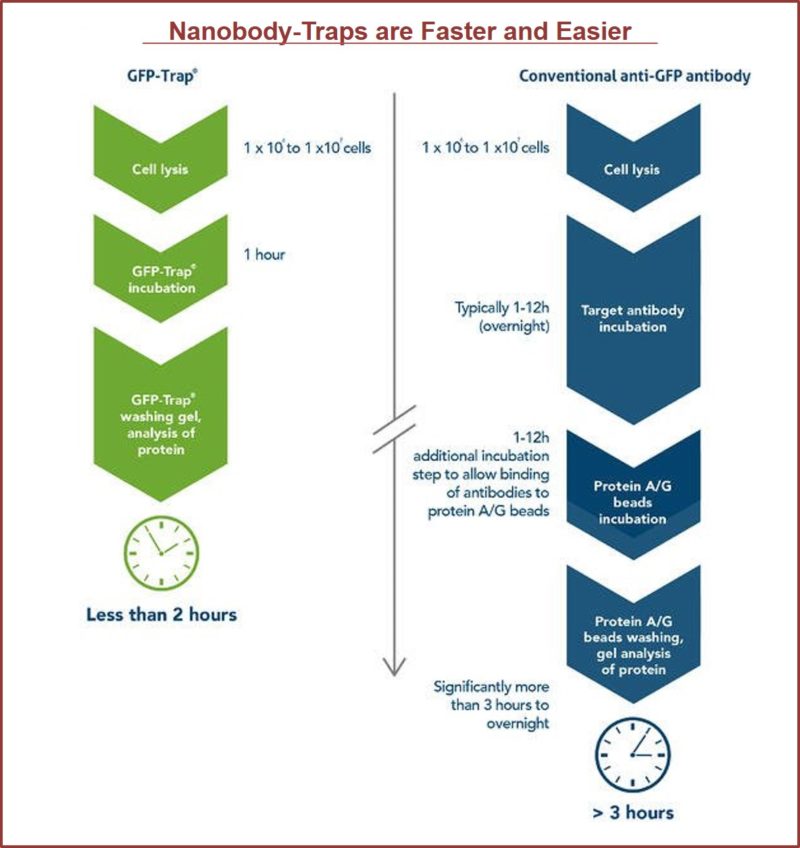
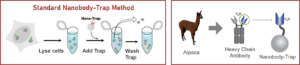
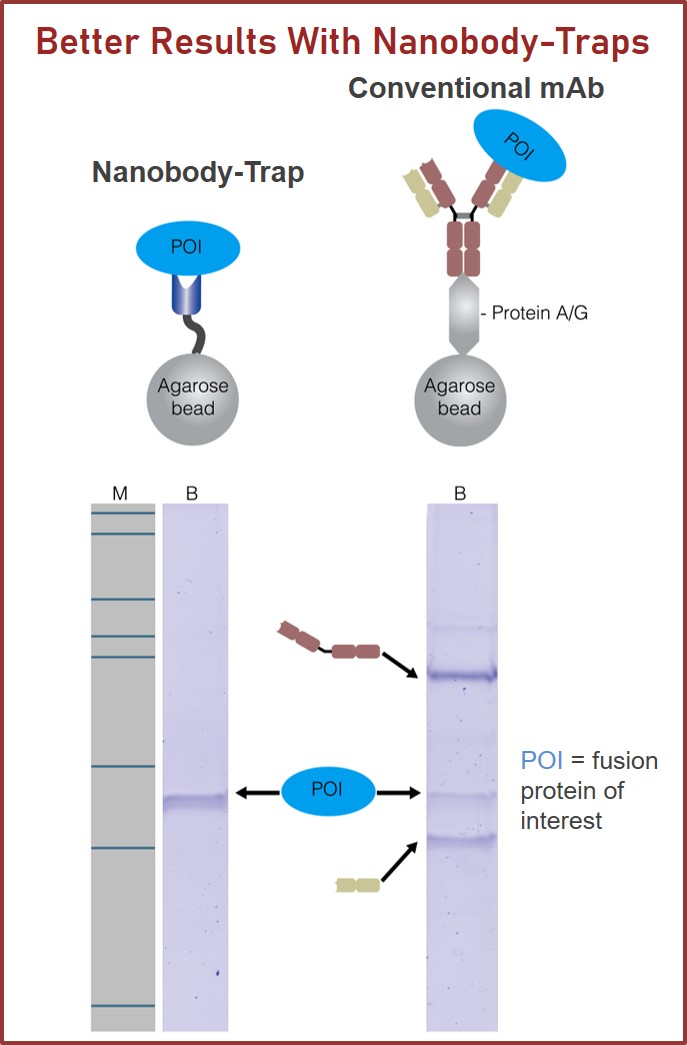
GFP-Traps utilize super-high affinity alpaca antibody fragments coupled to agarose beads or magnetic agarose beads. These “Nanobody-Traps” are perfect for immuno-precipitation, immuno-purification, and immuno-pull down experiments with up to 10-fold better purity (and yield) than that of conventional mouse monoclonal antibodies. Compatible with a variety of source materials, Nanobody-Traps may be used with mammalian cells, tissues & organs, bacteria, yeast, and even plants. These reagents allow your GFP-fusions to be perfect candidates for immunoprecipitations, Co-IP, mass spectroscopy, enzyme activity measurements, and ChIP analysis.
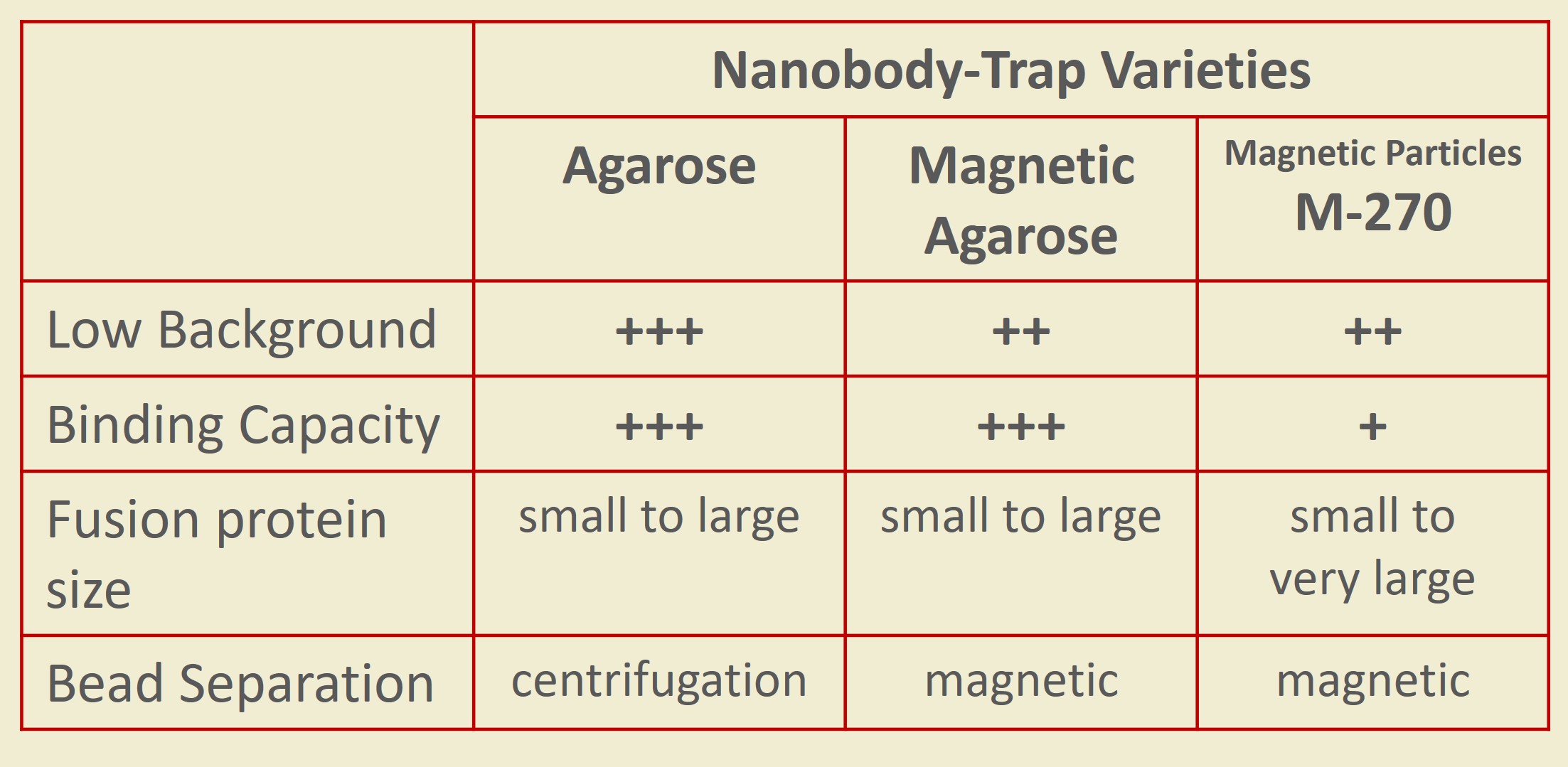
M-270 particles are made for the largest fusion proteins and large protein complexes!
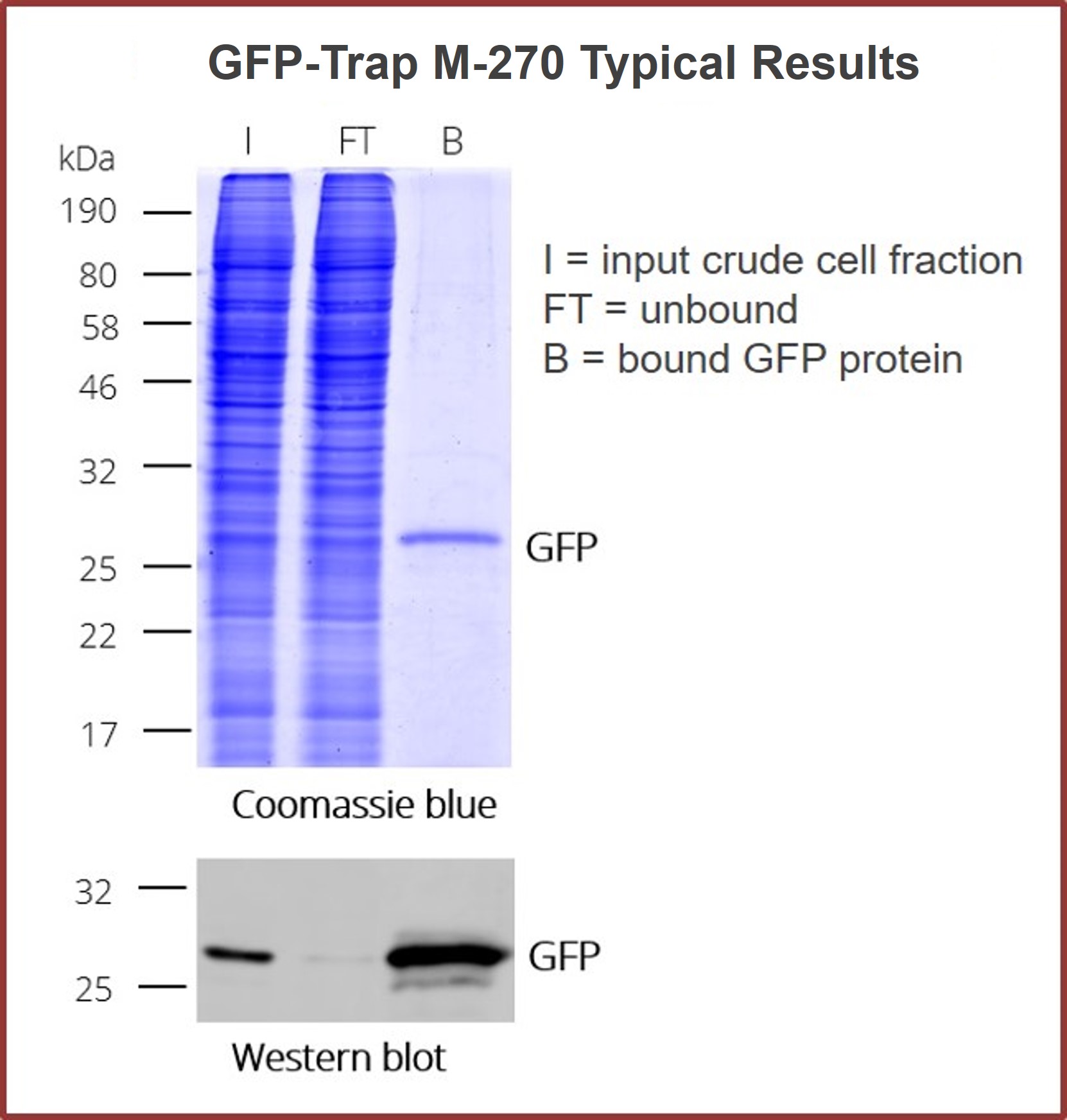
GFP-Trap Magnetic Particles M-270 are specifically designed to capture fusion proteins that are too big for our standard GFP-Trap Agarose or Magnetic Agarose beads. The larger and more consistently-sized M-270 provide plenty of room for loads of large GFP-fusion protein to bind on their surface. Just as fast and clean as the other GFP-Trap reagents — This version maintains a simple one-step isolation of your GFP fusion protein and its interacting factors, so your lab sacrifices nothing.
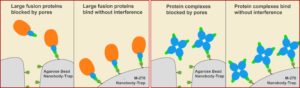
M-270 are non-porous, solid particles with ligands coupled on their surface. For this reason all sizes of GFP-tagged proteins — including very large GFP-tagged proteins (150+ kDA) — and multi-mers bind to GFP-Trap Magnetic Particle M-270. This immuno-purification reagent even work for protein complexes including: i) bulky proteins, and ii) secondary or tertiary partners. No matter what is bound, the beads can effectively immunoprecipitate under a number of conditions.

What’s your lab’s favorite flavor?
Nanobody-Traps are high quality binding proteins coupled to a monovalent matrix (agarose beads, magnetic particles, and the dynamite M-270 beads) for biochemical analysis of many fusion proteins and their interacting partners. The different formats allow you to perform a multitude of experiments. The most popular forms are Nanobody-Traps linked to agarose beads. Agarose beads provide the largest binding capacity and lowest background, while still being easy to work with. If automation is important to your lab, then magnetic agarose particles are a great choice. Choose Nanobody-Traps linked to M-270 beads for huge fusion proteins (over 150 kDa) or those that are part of a larger protein assembly. The purified protein complexes are compatible with wide range of down-stream analyses such as mass spectroscopy.
Over 50 fluorescent proteins have been checked with Nanobody-Traps, click to check!
Camelidae single-domain antibodies are like IgGs on steroids
The family of animals known as Camelidae (camels, llamas, and alpacas) produce functional antibodies devoid of light chains, so-called “heavy chain” antibodies. These heavy chain antibodies recognize and bind their antigens via a single variable domain. When cleaved from their carboxy tail, these barrel-shaped structures (2×3 nm) are extraordinarily small, naturally-occurring, and intact antigen binding fragments (MW of 13 kDa). These fragments, called Nanobodies, are characterized by high specificity, affinities in the low nanomolar range, and dissociation constants in the sub-nanomolar range (typically 10- to 100-fold better than mouse IgGs). The compact size of Nanobodies makes them extremely stable at temperatures up to 70°C, and functional even in 2M NaCl or 0.5% SDS. These small and powerful antibody fragments can be used in a variety of unique applications. They will open up your research possibilities.
Additional Information
“On my protein, which forms pentamers and also complexes with many other proteins, the GFP-Trap Dynabeads worked at least as well
as the agarose. Moreover, the manipulation with magnetic beads is much more easier.”
Researcher, Institute of Hematology and Blood Transfusion, Prague
“The new optimized GFP-Trap Dynabeads are indeed better than the GFP-Trap beads that we were using so far, for proteins larger than 200 kDa.”
Postdoctoral fellow, University of Groningen
“We compared the GFP-Trap Dynabeads with the MA [magnetic-agarose] beads and the Dynabeads are far superior not only for the amount of bait pull-down, but importantly also of its interaction partner.”
Lecturer in Cell Biology, University of Cambridge
“I’ve tried GFP-Trap Dynabeads sample that I received with my last order and it’s working perfectly!”
PhD student, Institute Curie, France
GFP-Traps Coupled to M-270 Magnetic Particles
|
SPECIFICATIONS
|
|
|---|---|
| Affinity | Dissociation constant KD of 1 pM |
| Part Numbers | GTD010, GTD020, GTD100, GTD200, GTD400, GTDK020, GT250 |
| Wash buffer compatibility | 10 mM DTT, 8 M Urea, 2 M NaCl, 2 % Nonidet P40 Substitute, 0.2 % SDS, 1 % Triton X-100 |
| Binding capacity | 10 µL slurry bind about 1 µg of recombinant GFP |
| Coupled Nanobody/ VHH | Monoclonal anti-Green Fluorescent Protein (GFP) single domain antibody (sdAb) fragment, recombinantly expressed |
| Specificity | – AcGFP, Clover, eGFP, Emerald, GFP, GFP5, GFP Envy, GFP S65T, mGFP, mPhluorin, PA-GFP, Superfolder GFP, TagGFP, TagGFP2, monomeric eGFP A206K – CFP – YFP, Citrine, eCitrine, eYFP, Venus, Ypet – BFP |
| Elution | – SDS sample buffer – 0.2 M glycine pH 2.5 Instead of elution, we recommend on-bead assays like on-bead digestion for MS analysis. |
| Particle Size | ~2.8 µm |
| Compatibility with mass spectrometry | The GFP-Trap is optimized for on-bead digestion. Complete tryptic digest results in 4-5 peptides. |
| Storage Buffer | 1x PBS; Preservative: 0.09% Sodium Azide |
| Storage and Stability | store at 4°C; stable for one year. Do not freeze. |



