MIDORI Green Direct — Safe DNA Stain
Add to sample before loading
Put it in your sample not your gel
MIDORI Green Direct is a specially constructed fluorescent molecule that you add to your sample, not into the gel or the buffer. It will bind and label nucleic acid fragments (DNA or RNA) as they separate and resolve during a standard electrophoresis run. These labeled molecules will shine brightly against a near-black background. By comparison, nucleic acid stains that are added to molten agarose (before the gel is cast) often have a “two-tone look” as free dye moves in the reverse direction of the DNA fragments and causes the bottom of the gel to be much darker than the top. Many labs prefer MIDORI Green Direct when sensitivity of detection or ease of use are important considerations.

Comparison of sensitivity between MIDORI Green Direct (left green) using a Blue/Green LED illuminator and Ethidium Bromide (right red) using a UV Transilluminator.
MIDORI Green Direct is provided in a format of 10X sample loading dyes and is designed to be added “directly” to your samples. You do not need to add it, or any other dyes, to the gel matrix or to the running buffers. A 1 mL tube is enough for 1,000 to 2,000 samples (lanes).
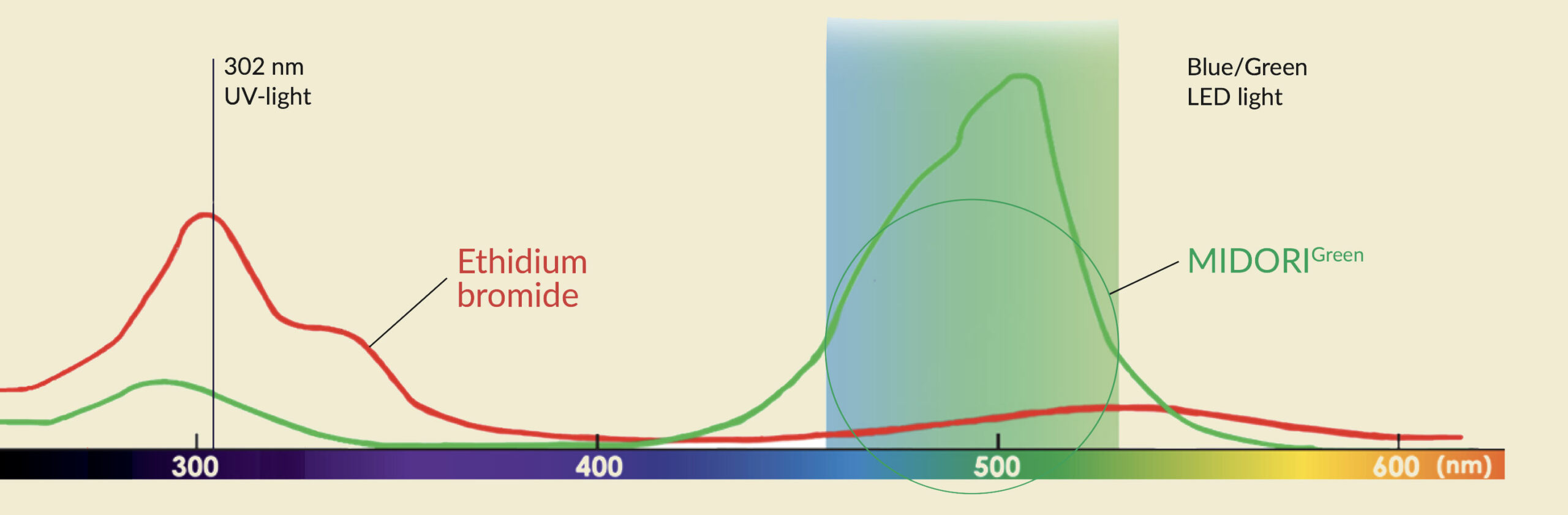
The absorbance spectrum of Ethidium Bromide, MIDORI Green Advance, and MIDORI Green Direct
Not only can you use your existing UV transilluminator and filter sets — MIDORI Green Direct works best when matched with blue LED-, or in particular, Blue/Green LED-based illuminators and gel documentation systems. These environmentally-friendly light tables are the perfect complement for any MIDORI Green stain, as you can work shield-less and goggle-less when excising bands or imaging migration. And because blue or green light does not cross-link DNA, you never need to worry about damaging DNA during cloning and photographing sessions.
Thoroughly tested for your safety
Non-toxic. Non-mutagenic. MIDORI Green Direct Nucleic Acid Stain is a safe alternative to traditional ethidium bromide(EtBr) stain for detecting dsDNA, ssDNA and RNA in agarose gels. This proprietary stain has been documented to be safe in by four different tests. MIDORI Green Direct causes fewer mutations in the Ames Tests, the accepted industry standard for carcinogen and teratogen testing. High mutagenicity in this test is often linked to carcinogens and teratogens. Another side of safety is toxicity, and all Midori Green stains are non-toxic too! The unique molecular structure of MIDORI Green Direct makes it unable to pass through the plasma membrane of living cells. This allows an added layer of protection so that Direct cannot even make to the DNA or RNA molecules the reside within an organism’s cells. Both Aquatic Toxicity Testing and Corrosivity, Reactivity and Ignitability Testing show MIDORI Green Direct not to be hazardous waste and can be disposed of according to standard laboratory procedures, making use in the lab easy as well as safe for technical staff and students. EtBr and some other “safe DNA dyes” are not consider to be non-toxic, and must be handled in a more rigorous fashion. And for those who prefer to be extra safe, MIDORI Green Direct, like Advance, also tests negative for latex/nitrile glove penetration.
Three makes a happy family
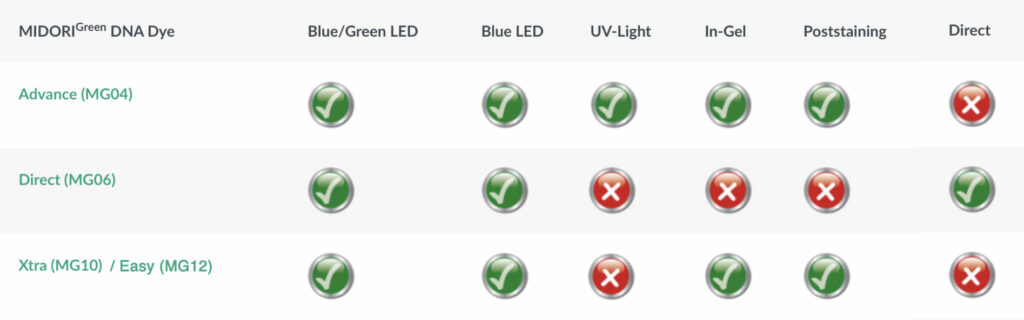
The core MIDORI Green molecule has been formulated into three different green fluorescent stains optimized for your lab’s needs. First, there’s MIDORI Green Advance, which offers an excellent signal-to-noise ratio and solid sensitivity for even short nucleic acid fragments. MIDORI Green Advance is added into the agarose gel prior to running samples (much like EtBr), and provides sensitivity of detection on par with EtBr when visualized with blue LEDs or Nippon Genetics’ superior Blue/Green LED technology. The second, MIDORI Green Direct, is designed to mix with your sample and load onto a stain-free gel. MIDORI Green Direct has the loading dye included, and detects nucleic acid fragments to a similar, if not slightly better, level as MIDORI Green Advance. The newest member of the family is MIDORI Green Xtra/Easy. Like Advance, Xtra/Easy is added to molten agarose and buffer. It’s chemical structure is optimized for Blue/Green and Blue LED light, leading to unbeatable fluorescence signals of DNA and RNA in agarose gels. MIDORI Green Advance and MIDORI Green Direct dyes are compatible with UV-light, but slightly less efficient when using UV rather than the non-damaging visible excitation light of LEDs. Most importantly for labs, MIDORI Green Xtra/Easy does not stain the agarose gel, leading to an excellent signal-to-noise ratio. Xtra/Easy makes the detection of even the most minute quantities of DNA or RNA possible (and certainly better than EtBr)—it’s not just a replacement for EtBr, it’s an upgrade.
Safer for you and better for your subcloning
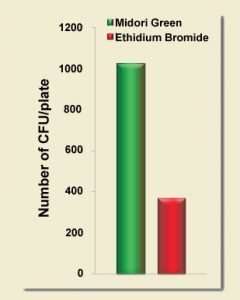
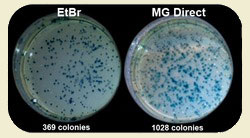
By using safe MIDORI Green dyes and safe Blue/Green LED illumination you can improve your subcloning transformation efficiencies by THREE-FOLD. In the example below, a plasmid vector was double digested with suitable restriction enzymes to create two sticky-ended DNA fragments: the lacZ gene (3,536 bp) and the backbone of the vector (4,318 bp). Equal amounts of digested DNA were electrophoresed on 1% agarose gels. The gels were stained with either ethidium bromide or MIDORI Green Direct gel stain according to the corresponding manuals, and then viewed using either a UV transilluminator or the FastGene Blue/Green LED Illuminator, respectively. The two DNA fragments were excised from the gels and purified using a silica membrane based purification kit. The lacZ gene and the vector backbone were re-ligated using T4 DNA ligase transformed into DH5a cells and plated onto selection plates. The total number of blue and white colonies was counted to evaluate cloning efficiency. Each experiment was conducted in triplicate, and the average cloning efficiency was determined. MIDORI Green Direct resulted in a dramatic increase of positive transformants.
Midori Green Can Boost Your Cloning Results!
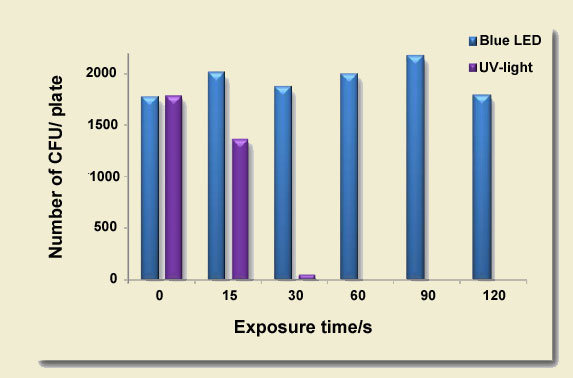
Ethidium bromide is typically used in conjunction with a strong UV light source to excise DNA bands for purification prior to the ligation reaction. Short-wavelength light is known to cause thymine dimers and damage DNA. The extent of this damage is not always appreciated. High-energy light wreaks havoc on DNA fragments in mere seconds. As can be seen below, cloning efficiency starts to drop after just a 15 second exposure of DNA in a standard agarose gel. After a 30 second exposure, your cloning experiment is all but dead! In contrast, the cloning efficiency of protocols that use blue LEDs or Nippon Genetics’ super-performing Blue/Green LEDs are completely unaffected by this deleterious effect. If your lab can’t to break itself of its ethidium bromide habit, using a Blue/Green LED Illuminator (or imaging system) should still have an immediate positive impact on DNA integrity and cloning efficiency.
UV Transilluminators Kill Cloning Experiments

Midori Green Direct DNA Stain
|
SPECIFICATIONS
|
|
|---|---|
| Part Numbers | MG06 |
| Contents | Midori Green DNA Stain, Xylene Cyanol FF, and Orange G at 10x conc. |
| DMSO | Yes |
| Add to Gel | No |
| Add to Sample | Yes |
| Post Staining | No |
| UV Transilluminator Compatible | Yes Recommended: 300 nm – 310 nm Not recommended: 254 nm |
| Blue LED Illuminator Compatible | Yes |
| Blue/Green LED Illuminator Compatible | Yes, best choice |
| Excitation | 270 nm / 500 nm |
| Emission | 530 nm |
| Toxicity | Non-toxic Non-carcinogenic Non-mutagenic |
| Storage | 1 Year in the dark at 4°C |


