Spot-Trap Particles M-270 Affinity Reagents
Capture large Spot-tagged Proteins
Simple purification with ultra-low background
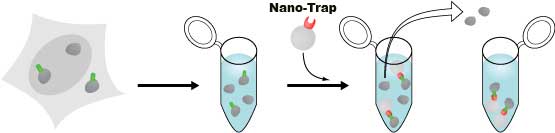
The Spot-Trap is a high quality Spot-Tag binding protein coupled to a resin matrix designed for use with immunoprecipitation or affinity purification protocols. It will only interact with the novel 12 amino acid sequence found on Spot-Tag fusion proteins. Spot-Tag has many advantages as compared with other short peptide “tag” systems. Like all Nanobody-Traps, the Spot-Trap binding protein is a small (14.7 kDa), monovalent, high-affinity monoclonal alpaca antibody (“nanobody”), and perfectly recognizes the Spot-Tag sequence motif (PDRVRAVSHWSS) at either the N-terminus or C-terminus of the expressed fusion protein. The Spot-Trap is now offered covalently coupled to the surface of Particles M-270 , or agarose or magnetic-agarose beads for fast and effective immunoprecipitation and affinity purification. It is optimized for biochemical analysis of Spot-Tagged proteins and their interacting partners. It has been tested for multiple applications and works efficiently in various systems, such as bacteria, yeast, mammalian cell lines, and insect cells. For biochemical analysis including mass spectrometry and enzyme activity measurements, Spot-Tag-fusion proteins and their interacting factors can be isolated quickly and efficiently by immunoprecipitation using the Spot-Trap.

M-270 particles are made for the largest fusion proteins and large protein complexes!
Spot-Trap M-270 particles are specifically designed to capture fusion proteins that are too big for our standard Spot-Trap Agarose or Magnetic Agarose beads. The larger and more consistently-sized M-270 particles provide plenty of room for loads of large Spot-fusion protein to bind on their surface. Just as fast and clean as the other Spot-Trap reagents — The M-270 particle version maintains a simple one-step isolation of your Spot fusion protein and its interacting factors, so your lab sacrifices nothing.
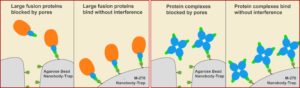
M-270 particles are non-porous, solid particles with ligands coupled on their surface. For this reason all sizes of Spot-tagged proteins — including very large SPOT-tagged proteins — and multi-mers bind to Spot-Trap M-270 particles. This immuno-purification reagent even work for protein complexes including: i) bulky proteins, and ii) secondary or tertiary partners. No matter what is bound, the beads can effectively immunoprecipitate under a number of conditions.

What’s your lab’s favorite flavor?
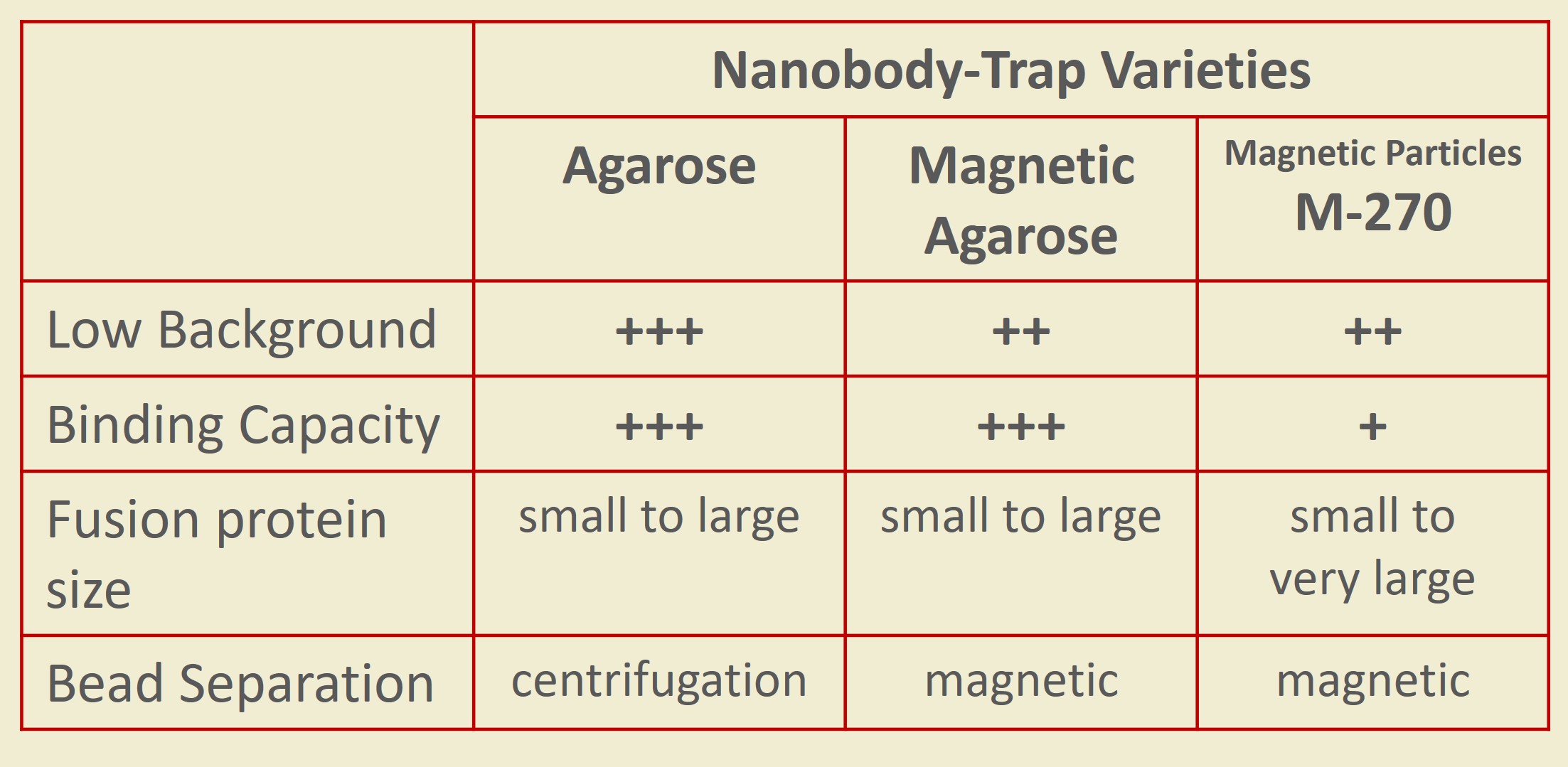
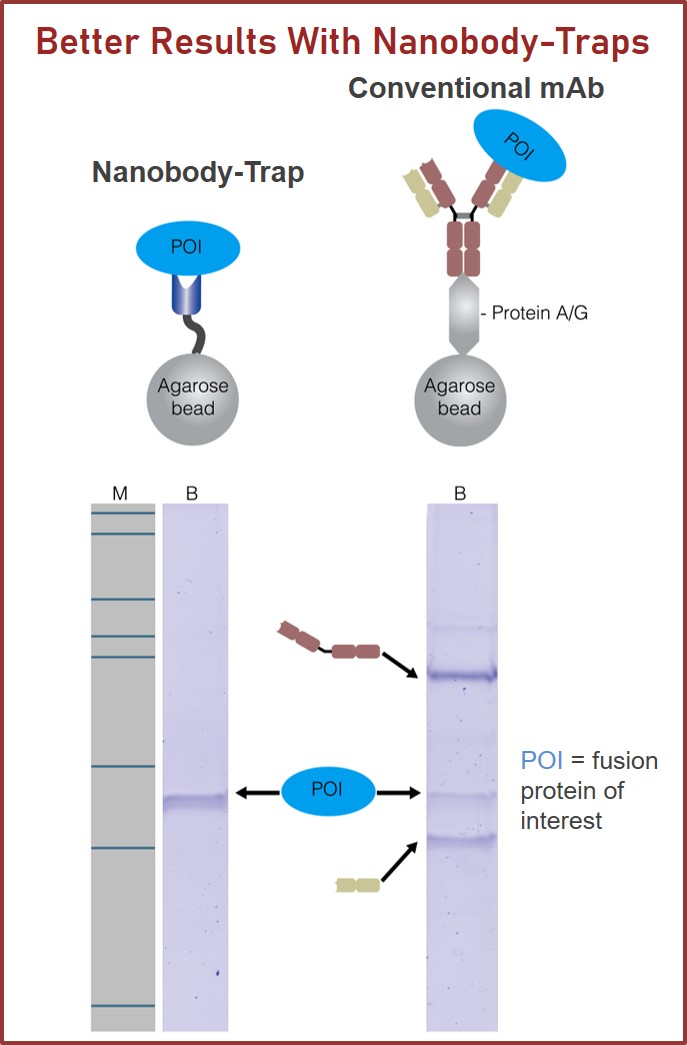
Nanobody-Traps are high quality binding proteins coupled to a monovalent matrix (agarose beads, magnetic particles, and the dynamite M-270 beads) for biochemical analysis of many fusion proteins and their interacting partners. The different formats allow you to perform a multitude of experiments. The most popular forms are Nanobody-Traps linked to agarose beads. Agarose beads provide the largest binding capacity and lowest background, while still being easy to work with. If automation is important to your lab, then magnetic agarose particles are a great choice. Choose Nanobody-Traps linked to M-270 beads for huge fusion proteins (over 150 kDa) or those that are part of a larger protein assembly. The purified protein complexes are compatible with wide range of down-stream analyses such as mass spectroscopy.
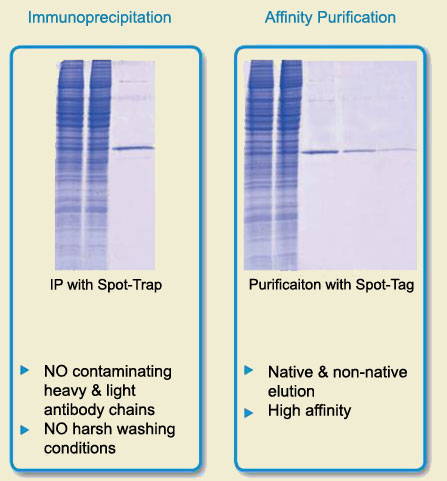
When the results matter use Spot-Trap
To the left, immunoprecipitation was performed on a Spot-Tagged GFP. It is apparent that Spot-Trap not only lacks heavy and light chain antibody contaminations, but also lacks non-specific binding of contaminating proteins. This makes Spot-Trap a superior affinity capture tool for single band purification of Spot-tagged proteins. To the right, the same Spot-Tagged GFP was bound to matrix and then competitively eluted with 100 µM Spot-Peptide. This gentle affinity purification technique is a perfect way to preserve protein structure and function, and to look at larger protein complexes.
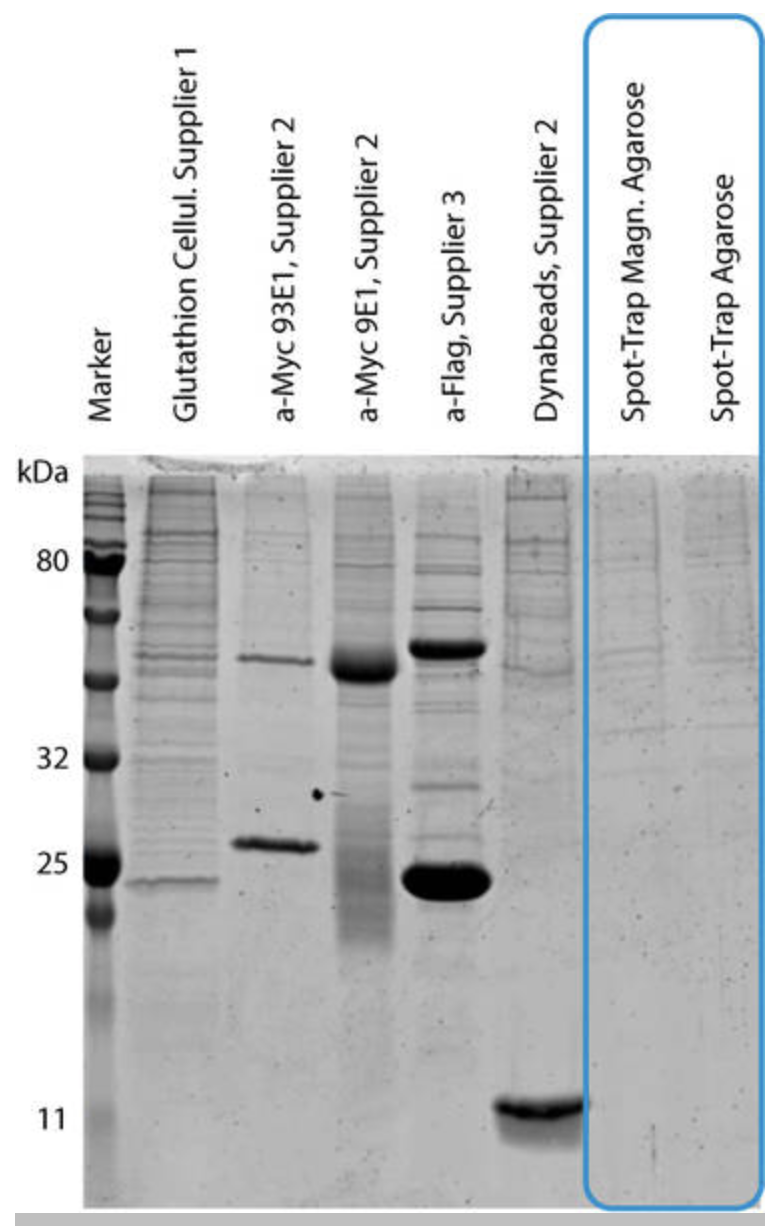
Spot-Trap is specially designed for spotless background
To the right, immunoprecipitations were conducted with HEK293T cell lysates without Spot-Tag or any other expressed tagged proteins present (each lane was normalized for equal binding capacity). The IPs were performed according to respective manufacturers’ protocols. Both versions of Spot-Trap affinity reagent show almost no non-specific binding of endogenous mammalian cell proteins because of a superior, re-engineered wash buffer. This promises clean results every time!
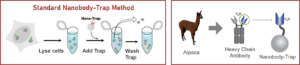
Camelidae single-domain antibodies are like IgGs on steroids
The family of animals known as Camelidae (camels, llamas, and alpacas) produce functional antibodies devoid of light chains, so-called “heavy chain” antibodies. These heavy chain antibodies recognize and bind their antigens via a single variable domain. When cleaved from their carboxy tail, these barrel-shaped structures (2×3 nm) are extraordinarily small, naturally-occurring, and intact antigen binding fragments (MW of 13 kDa). These fragments, called Nanobodies, are characterized by high specificity, affinities in the low nanomolar range, and dissociation constants in the sub-nanomolar range (typically 10- to 100-fold better than mouse IgGs). The compact size of Nanobodies makes them extremely stable at temperatures up to 70°C, and functional even in 2M NaCl or 0.5% SDS. These small and powerful antibody fragments can be used in a variety of unique applications. They will open up your research possibilities.
Spot-Traps Coupled to M-270 Magnetic Particles
ETD010 – Spot-Trap, coupled to Particles M-270 , 10 rxns, 0.25 ml
Original price was: $352.00.$340.00Current price is: $340.00. Add to cart
ETD020 – Spot-Trap, coupled to Particles M-270 , 20 rxns, 0.5 ml
Original price was: $578.00.$565.00Current price is: $565.00. Add to cartSpot-Trap Non-Coupled
Spin Columns for agarose beads
|
SPECIFICATIONS
|
|
|---|---|
| Configuration: | Recombinant, monoclonal anti-Spot-Tag single domain antibody (sdAb) fragment coupled to Particles M-270 |
| Specificity: | Spot-Tag sequence motif PDRVRAVSHWSS |
| Particle Size: | 2.8 µm |
| Binding Capacity: | 10µl slurry binds about 0.9 µg of recombinant Spot-tagged protein (30 kDa) |
| Storage Buffer: | 1 x PBS, Preservative: 0.09% sodium azide |
| Washing buffer compatibility: | 2 M NaCl, 10 mM DTT, 2 % Nonidet P40 Substitute, 2 % Triton X-100 |
| Part Numbers | ETD010, ETD020, ETD100, ETD200,ETD400, ETDK020, ep1, etb250 |
| Storage and Stability: | store at 4°C; stable for one year. Do not freeze. |



